
Sulfuric Acid
Sulfuric acid is often called “the backbone of the chemical industry“. It is one of the most widely used chemicals in the world with applications spanning the production of fertilizers, batteries, viscose fibers, pulp and paper, pigments, pharmaceuticals, metal processing, as well as the manufacture of other industrial chemicals, like caprolactam or hydrofluoric acid.
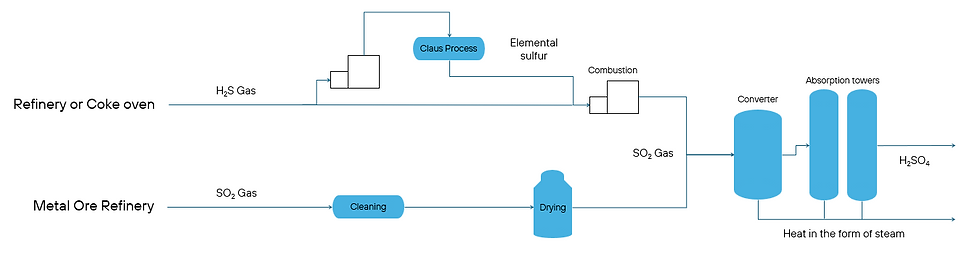
The main feedstock for sulfuric acid production is sulfur recovered from coking, and crude oil or natural gas refining in the form of hydrogen sulfide gas (H₂S) or in the form of solid or liquid elemental sulfur (Sₓ) from the Claus process, for better transportation. H₂S or elemental sulfur is then burned in a combustor to produce sulfur dioxide gas (SO₂). For Claus process, Envitrack supplies RCAT®-9000 series for high efficiency sulfur recovery.
Second largest source of sulfur is off-gas from processing of sulfide metal ores, especially copper, zinc, nickel, cobalt or iron. Here, the sulfur is already in the form of SO₂ gas, which first needs to be cleaned from impurities. Depending on the following process, the gas can then be dried, thermoregulated to the desired temperature and fed into the catalytic converter.
Here our catalyst from RCAT®-7200 series transforms sulfur dioxide into sulfur trioxide by reacting it with airborne oxygen. If the incoming gas is dried and Contact process is being utilized, the generated sulfur trioxide gas is then fed through Absorption towers, where it reacts with water to form sulfuric acid. Using variations of this process, oleum (or fuming sulfuric acid) can also be produced.
A different configuration called Wet-Gas Sulfuric Acid process can also be used. The incoming gas is maintained at a defined humidity and the formed SO₃ reacts with moisture in the air to generate sulfuric acid in the gaseous phase. This gas then passes through Condensation towers, where the acid condenses into liquid form and can be further diluted to desired concentrations.

Both processes create a lot of excess thermal energy. Heat exchangers are utilized to recover this heat in the form of superheated steam and to control the temperature in the catalytic converter. Different configuration and number of Converter beds, Heat exchangers and Absorption or Condensation towers are possible for both processes.
One of the common solutions is Double Contact Double Absorption process.
Here the feedstock is fed over 2 out of 4 catalytic Converter beds, then through a Heat exchanger and one of two Absorption towers, to reduce the amount of SO₃ and help move the equilibrium towards the desired products. Afterwards the intermediate gas is fed back into the converter to react the remaining SO₂ to SO₃ and through the second Absorption tower. This configuration enhances the conversion to sulfuric acid and maximizes heat utilization of the whole plant.
Similar solution can be used for the Wet-Gas Sulfuric Acid process. The gas from the second converter bed can be fed through a Heat exchanger and the first Condensation tower, recovering part of the produced sulfuric acid and also absorbing some amount of SO₃ to move the equilibrium towards the desired product. Intermediate gas is then fed back over the remaining Converter beds and the final pass through the second Condensation tower to recover the remaining acid and recuperate excess heat.
Typical industrial solutions can achieve over 99.5 % SO₂ to SO₃ conversion and produce acid of varying concentration.
Sulfuric Acid Production Catalysts
-
WSA Process catalyst RCAT®-7240
-
Cesium promoted WSA Process catalyst RCAT®-7241
-
Contact Process catalyst RCAT®-7250
-
Cesium promoted Contact Process catalyst RCAT®-7252
